A denitrogenative palladium-catalyzed cascade for regioselective synthesis of fluorenes†
Abstract
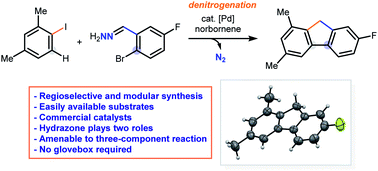
We herein report a denitrogenative palladium-catalyzed cascade for the modular and regioselective synthesis of polysubstituted fluorenes. Hydrazone facilitates the Pd(II) to Pd(IV) oxidative addition in a Catellani pathway and is also the methylene synthon in the proposed reaction. Aryl iodides and 2-bromoarylaldehyde hydrazones undergo a norbornene-controlled tandem reaction sequence to give a broad scope of fluorenes in the presence of a palladium catalyst. The method described is scalable and adaptable to a three-component reaction with in situ generation of the hydrazone group. Preliminary mechanistic investigations have been conducted.


 Please wait while we load your content...
Please wait while we load your content...