Integrated multi-step continuous flow synthesis of daclatasvir without intermediate purification and solvent exchange†
Abstract
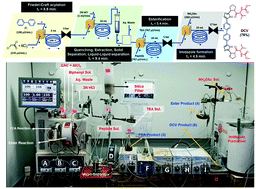
The rapid transmission of viral diseases can cause massive economic damage and loss of life. The manufacture of most anti-viral drugs is normally carried out using batch synthesis which typically requires long production times (3–15 days) and significant manpower and infrastructure. The fabrication of fully integrated flow-based manufacturing systems offers an alternative approach that can be greener and more economical. In this regard, we have developed a compact manufacturing machine for the synthesis of the antiviral API daclatasvir which is easily reconfigured, has a much-reduced footprint, and enables multi-step synthesis using innovative reaction chemistry and post-synthesis purification equipment. The developed integrated system enabled the ultra-fast production of DCV as its free base (within 28.2 min.) with a throughput of 11.8 g per day (equivalent to 200 doses of Daklinza, 60 mg tablets) in 98% HPLC purity.


 Please wait while we load your content...
Please wait while we load your content...