Kinetic modelling of hydrogen transfer deoxygenation of a prototypical fatty acid over a bimetallic Pd60Cu40 catalyst: an investigation of the surface reaction mechanism and rate limiting step†
Abstract
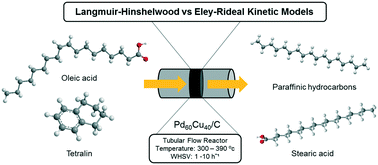
Herein, for the first time, we demonstrate a novel continuous flow process involving the application of tetralin as a hydrogen donor solvent for the catalytic conversion of oleic acid to diesel-like hydrocarbons, using an efficient and stable carbon-supported bimetallic PdCu catalyst. Using Pd60Cu40/C, where 60 : 40 is the molar ratio of each metal, at optimum reaction conditions (360 °C and WHSV = 1 h−1), 90.5% oleic acid conversion and 80.5% selectivity to C17 and C18 paraffinic hydrocarbons were achieved. Furthermore, a comprehensive mechanistic based kinetic modelling – considering power rate law, L–H and E–R models was conducted. Kinetic expressions derived from the three kinetic models were investigated in rate data fitting through nonlinear regression using a Levenberg–Marquardt algorithm. Based on the statistical discrimination criteria, the experimental data of the dehydrogenation reaction of tetralin were best fitted by an L–H rate equation assuming the surface reaction as the rate controlling step. In contrast, the kinetic data of the oleic acid deoxygenation reaction were well correlated with an L–H rate equation assuming single site adsorption of oleic acid with dissociative H2 adsorption. It was found that the rate limiting step of the overall reaction was the hydrogenation of oleic acid with an activation energy of 75.0 ± 5.1 kJ mol−1 whereas the dehydrogenation of tetralin had a lower activation energy of 66.4 ± 2.7 kJ mol−1.


 Please wait while we load your content...
Please wait while we load your content...