Homogeneous synthesis of hydroxylamine hydrochloride via acid-catalyzed hydrolysis of nitromethane†
Abstract
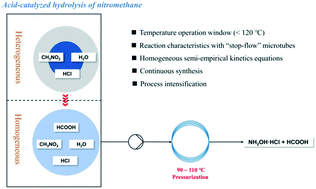
The acid-catalyzed hydrolysis of nitromethane takes advantage of producing high-quality hydroxylamine hydrochloride, an important chemical material, but also draws risk concerns in scale up and batch operation of the heterogeneous process. To resolve these problems, a homogeneous synthesis system was established in this work by adding the by-product formic acid to the reaction mixture. Based on the homogeneous system, a stop-flow microtube reactor was used to investigate the hydrolysis reaction characteristics and to develop the corresponding semi-empirical kinetics equation. Kinetics experiments were performed at less than 120 °C in order to suppress side reactions. We elucidated that water and acid imposed complex effects on the hydrolysis reaction rate in two separate intervals of acid concentration. Kinetics models were established in these two regions respectively and provided an excellent correlation to the experimental data. The large overall activation energy indicates that the hydrolysis reaction is sensitive to reaction temperature. The intensified contact between reactants furnished by the homogeneous system, and optimized temperature (110 °C) and HCl concentration (>5.0 M) can significantly increase the reaction rate. Compared with that of the conventional process, the reaction time was greatly shortened to four hours for reaching 90% conversion of nitromethane at 110 °C. The high consistency of the stop-flow reactor and continuous flow reactor in reaction performances was experimentally confirmed, verifying the feasibility of the well-designed flow synthesis of hydroxylamine hydrochloride.

 Please wait while we load your content...
Please wait while we load your content...