Rapid CO2 capture-to-mineralisation in a scalable reactor†
Abstract
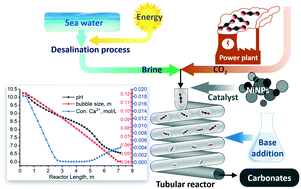
CO2 mineralisation is a process that can convert CO2 into solid carbonates for permanent storage. Our multiphase flow process uses an alkaline brine solution to capture gaseous CO2 and form carbonate particles in a continuous tubular reactor. In this research, a monoethanolamine (MEA) solution is utilised to synergistically boost CO2 solubility in the brine while neutralising the acidification caused by brine chloride ions left in the solution following the precipitation of alkaline earth metals. In this study, relatively low concentrations of MEA, ranging from 0.036 to 0.33 M, were investigated over a temperature range from 303 K to 323 K; these are significantly milder conditions than those used in traditional CO2 capture processes with MEA, which contributes to low energy demand of the process. Short residence time, in the order of a few minutes, is made possible by the high gas–liquid surface area for mass transfer and the rapid kinetics of aqueous phase carbonation reactions. Nickel nanoparticles (NiNPs) were tested as a catalytic additive to further accelerate the rate limiting step (CO2 dissolution) by accelerating the CO2 hydration reaction. Experimental results were used to develop and calibrate a one-dimensional time-dependent plug-flow model that incorporates transport and chemical speciation equations. The model is thus capable of predicting aqueous species and solid carbonate concentrations, fluid pressure and gas slug size as a function of reactor length. These in turn yield the carbonation conversion and total pressure drop, and provide mechanistic insight into the reactor processes that can be used for scale-up. The experimental and modelled results showed a good agreement for a wide range of conditions tested: effects of temperature, brine composition, MEA concentration, and gas–liquid flow ratio. Under optimum conditions, it was found that the reactor could achieve full conversion of calcium from the brine and CO2 from the gas phase, thus proving to be an efficient process with high atom economy.


 Please wait while we load your content...
Please wait while we load your content...