Modelling protein therapeutic co-formulation and co-delivery with PLGA nanoparticles continuously manufactured by microfluidics
Abstract
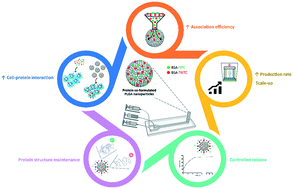
Formulating protein therapeutics into nanoparticles (NPs) of poly(lactic-co-glycolic acid) (PLGA) provides key features such as protection against clearance, sustained release and less side effects by possible attachment of targeting ligands. These NPs also offer the potential for protein combination therapy, which is expected to exploit synergetic bioresponses, avoid multiple dosage regimens and consequent mis-dosing. Since the conventional manufacture of protein-loaded PLGA NPs is still associated with low-throughput, new continuous manufacturing methods such as microfluidics have been established. Herein, PLGA NPs continuously manufactured through microfluidics are proposed for co-formulation and -delivery of two model proteins consisting of bovine serum albumin (BSA) conjugated to either fluorescein (FITC) or tetramethylrhodamine (TRITC) isothiocyanates. Protein co-formulated NPs of 100 nm, monodispersed and with 70% of association efficiency were obtained. The microfluidic setup allowed a production rate of around 7 g of particles per day and demonstrated scale-up capacity. Model proteins were released in a controlled manner and without significant changes in their secondary structure. Studies in macrophage-like cells proved that protein co-formulated PLGA NPs did not impair metabolic activity (>70%). The cellular association of the proteins was around 2-times higher when co-formulated into PLGA NPs, compared to the free protein controls. Moreover, the cellular association of the co-formulated proteins was 4-times higher than the physical mixture of NPs individually loaded with each protein type. This work has demonstrated the effectiveness of continuously manufactured PLGA NPs for co-formulating and enhancing the cellular association of co-delivered model proteins, providing a proof-of-concept foundation for future protein combination nanotherapies.


 Please wait while we load your content...
Please wait while we load your content...