Simultaneous catalytic reduction of SO2 and NO from flue gas using H2S as a reductant at low temperatures
Abstract
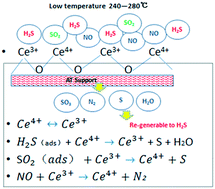
Although harmful NO and SO2 in flue gas can be separately removed by established technologies, such as selective catalytic reduction (SCR) or selective non-catalytic reduction (SNCR) and a wet scrubbing process, an integrated method for simultaneous desulfurization and denitrification is still in demand and attractive to improve energy efficiency and reduce investment and operation costs. Upon this, a novel sulfur-cycling integrated technology (simultaneous catalytic desulfurization and denitrification with H2S (H2S-SCDD)) has been proposed and is considered to be a profitable solution for the flue gas treatment. However, a high operation temperature (over 600 °C) is required in the H2S-SCDD process for the sulfur-cycling flue gas treatment as reported. To realize the H2S-SCDD process at low temperatures, different catalysts were prepared and tested, such as Al2O3–TiO2 (AT) loaded with transition metal oxides. The results show that a CeO2-loaded AT catalyst (Ce–AT) was the suitable catalyst to balance NO and SO2 removal by the H2S-SCDD process in the temperature range of 300–400 °C. Increasing Ce loading on AT significantly increased the NO conversion but had a slightly negative effect on SO2 reduction. With 15% Ce loading on the AT catalyst (Ce15–AT), the optimal temperature for the H2S-SCDD process was 240–280 °C. While at 280 °C, the SO2 and NO conversions were about 75% and 90%, respectively, implying the efficient and simultaneous reduction of NO and SO2. Upon this, the sulfur-cycling process is promising and attractive for flue gas treatment in industrial sectors.


 Please wait while we load your content...
Please wait while we load your content...