Cascade reaction engineering on zirconia-supported mesoporous MFI zeolites with tunable Lewis–Brønsted acid sites: a case of the one-pot conversion of furfural to γ-valerolactone†
Abstract
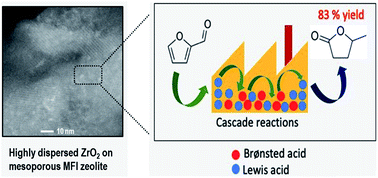
Catalytic cascade reactions are strongly desired as a potential means of combining multistep reactions into a single catalytic reactor. Appropriate catalysts composed of multi-reactive sites to catalyze cascade reactions in a sequential fashion are central to such efforts. Here, we demonstrate a bifunctional zeolite catalyst with close proximity of Brønsted and Lewis acid sites through the synthesis of a mesoporous ZrO2[Al]MFI nanosponge (NS). The unique mesopores of the MFI-NS allow the confinement of zirconium oxide clusters (Lewis acid sites, LA) within the few-unit-cell-thin MFI aluminosilicate zeolite wall (Brønsted acid sites, BA). Such a structure is clearly distinct from the conventional MFI zeolite, where the agglomeration of zirconium oxide clusters onto the external surface area within the crystal bulk is not possible, resulting in segregated BA and LA sites on the internal and external zeolite, respectively. By bringing the BA and LA within ZrO2[Al]MFI-NS 30, we uncovered a more efficient catalytic route for the conversion of furfural (100% within 2 h) to γ-valerolactone (GVL) (83%). This route is only evident when the long molecular diffusion path, in the most extreme case of physically mixed ZrO2-(LA) and Al-zeolites (BA) (45% of GVL yield), is eliminated. Unlike the bifunctional ZrO2–Al-beta (GVL yield of 75%), where the BA concentration is greatly compromised at the expense of LA formation, we also show that the ZrO2[Al]MFI-NS is able to maintain a high density and good stability of both types of acids.


 Please wait while we load your content...
Please wait while we load your content...