Facile synthesis of picenes incorporating imide moieties at both edges of the molecule and their application to n-channel field-effect transistors†
Abstract
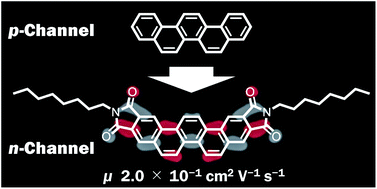
Picene derivatives incorporating imide moieties along the long-axis direction of the picene core (Cn-PicDIs) were conveniently synthesized through a four-step synthesis. Photochemical cyclization of dinaphthylethenes was used as the key step for constructing the picene skeleton. Field-effect transistor (FET) devices of Cn-PicDIs were fabricated by using ZrO2 as a gate substrate and their FET characteristics were investigated. The FET devices showed normally-off n-channel operation; the averaged electron mobility (μ) was evaluated to be 2(1) × 10−4, 1.0(6) × 10−1 and 1.4(3) × 10−2 cm2 V−1 s−1 for C4-PicDI, C8-PicDI and C12-PicDI, respectively. The maximum μ value as high as 2.0 × 10−1 cm2 V−1 s−1 was observed for C8-PicDI. The electronic spectra of Cn-PicDIs in solution showed the same profiles irrespective of the alkyl chain lengths. In contrast, in thin films, the UV absorption and photoelectron yield spectroscopy (PYS) indicated that the lowest unoccupied molecular orbital (LUMO) level of Cn-PicDIs gradually lowered upon the elongation of the alkyl chains, suggesting that the alkyl chains modify intermolecular interactions between the Cn-PicDI molecules in thin films. The present results provide a new strategy for constructing a high performance n-channel organic semiconductor material by utilizing the electronic features of phenacenes.


 Please wait while we load your content...
Please wait while we load your content...