Adsorption based realistic molecular model of amorphous kerogen
Abstract
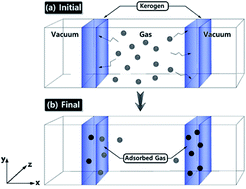
This paper reports the results of Grand Canonical Monte Carlo (GCMC)/molecular dynamics (MD) simulations of N2 and CO2 gas adsorption on three different organic geomacromolecule (kerogen) models. Molecular models of kerogen, although being continuously developed through various analytical and theoretical methods, still require further research due to the complexity and variability of the organic matter. In this joint theory and experiment study, three different kerogen models, with varying chemical compositions and structure from the Bakken, were constructed based on the acquired analytic data by Kelemen et al. in 2007: 13C nuclear magnetic resonance (13C-NMR), X-ray photoelectron spectroscopy (XPS), and X-ray absorption near-edge structure (XANES). N2 and CO2 gas adsorption isotherms obtained from GCMC/MD simulations are in very good agreement with the experimental isotherms of physical samples that had a similar geochemical composition and thermal maturity. The N2/CO2 uptake by the kerogen model at a range of pressure shows considerable similarity with our experimental data. The stronger interaction of CO2 molecules with the model leads to the penetration of CO2 molecules to the sub-surface levels in contrast to N2 molecules being concentrated on the surface of kerogen. These results suggest the important role of kerogen in the separation and transport of gas in organic-rich shale that are the target for sequestration of CO2 and/or enhanced oil recovery (EOR).


 Please wait while we load your content...
Please wait while we load your content...