Palladium nanoparticles on a pyridinium supported ionic liquid phase: a recyclable and low-leaching palladium catalyst for aminocarbonylation reactions†
Abstract
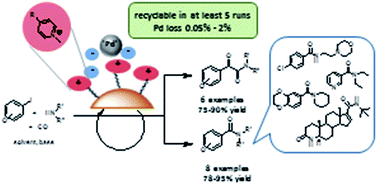
A new SILP (Supported Ionic Liquid Phase) palladium catalyst was prepared and characterized by 13C and 29Si CP MAS NMR, DTG, FTIR and TEM. The presence of the grafted pyridinium cations on the surface of the support was found to result in the formation of highly dispersed Pd nanoparticles with their diameter in the range of 1–2 nm. The catalyst was proved to be active not only in the aminocarbonylation of some model compounds but also in the synthesis of active pharmaceutical ingredients. Catalyst recycling and palladium leaching studies were carried out for the first time in aminocarbonylations leading to CX-546(1-(1,4-benzodioxan-6-ylcarbonyl)piperidine), Moclobemide, Nikethamide and a precursor of Finasteride. The latter reaction proves that not only aryl iodides but also an iodoalkene can be converted into the products with the help of the heterogeneous catalyst. The results show that the conditions should be always fine-tuned in the reactions of different substrates to achieve optimal results. Palladium loss was also observed to depend considerably on the nature of the reaction partners.


 Please wait while we load your content...
Please wait while we load your content...