Unbound water in mechanochemical reactions of brown coal
Abstract
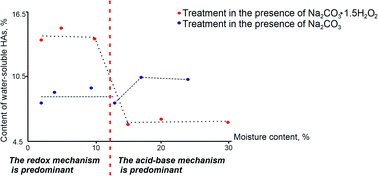
Mechanochemical activation of coal is commonly employed in industry. However, even the simplest solid-phase reactions, such as neutralization of humic acids in brown coal, remain insufficiently studied. The hypothesis regarding the occurrence of mechanochemical neutralization under local hydrothermal conditions for humic acids in brown coal has been tested in this study. 3D modelling of the “block–interlayer” system (where coal particles are separated by air interlayers saturated with water vapor) was used. The 3D model showed that the permittivity is expected to rise from 14 to 16% as the moisture content in the system increases from 12 to 15%. The actual permittivities of coal with different moisture contents have been measured by dielectric spectroscopy. In the real system, the permittivity increases more than threefold as the moisture content rises from 12 to 15%. This increase is much greater than the calculated one, demonstrating that the phase containing unbound water appears in the system at a moisture content of ∼12–13% and may exert various effects on the solid-phase reaction. There is a correlation between the moisture content, permittivity, and predominant mechanisms of the reaction between the organic matter in brown coal and sodium percarbonate (a reagent simultaneously containing the alkaline and peroxidic components). The reactions between brown coal and alkaline reagents proceed under local hydrothermal conditions. Both the alkaline and peroxidic components of sodium percarbonate participate in the solid-phase reaction between brown coal and sodium percarbonate. The emergence of unbound water in coal significantly inhibits the oxidation reaction.


 Please wait while we load your content...
Please wait while we load your content...