Alkaline water-splitting reactions over Pd/Co-MOF-derived carbon obtained via microwave-assisted synthesis
Abstract
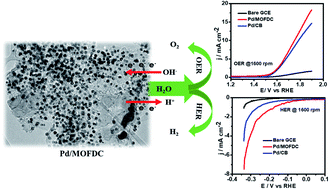
Cobalt-based metal–organic framework-derived carbon (MOFDC) has been studied as a new carbon-based support for a Pd catalyst for electrochemical water-splitting; i.e., the hydrogen evolution reaction (HER) and oxygen evolution reaction (OER) in alkaline medium. The study shows a high increase in the HER activity, in terms of low onset overpotential (onset η = 35 mV vs. RHE), high exchange current density (jo,s ≈ 0.22 mA cm−2), high mass activity (jo,m ≈ 59 mA mg−1), high kinetic current (jK ≈ 5–8 mA cm−2) and heterogeneous rate constant (k0 ≈ 4 × 10−4 cm s−1), which are attributed to the high porosity of MOFDC and contribution from residual Co, while the large Tafel slope (bc = 261 mV dec−1) is ascribed to the high degree of hydrogen adsorption onto polycrystalline Pd as a supplementary reaction step to the suggested Volmer–Heyrovsky mechanism. These values for the catalyst are comparable to or better than many recent reports that adopted nano-carbon materials and/or use bi- or ternary Pd-based electrocatalysts for the HER. The improved HER activity of Pd/MOFDC is associated with the positive impact of MOFDC and residual Co on the Pd catalyst (i.e., low activation energy, EA ≈ 12 kJ mol−1) which allows for easy desorption of the Hads to generate hydrogen. Moreover, Pd/MOFDC displays better OER activity than its analogue, with lower onset η (1.29 V vs. RHE) and ba (≈78 mV dec−1), and higher current response (ca. 18 mA cm−2). Indeed, this study provides a new strategy of designing and synthesizing MOFDC with physico-chemical features for Pd-based electrocatalysts that will allow for efficient electrochemical water-splitting processes.


 Please wait while we load your content...
Please wait while we load your content...