Coordination environment evolution of Co(ii) during dehydration and re-crystallization processes of KCoPO4·H2O towards enhanced electrocatalytic oxygen evolution reaction†
Abstract
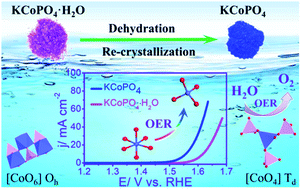
Development of efficient and stable electrodes for electrocatalytic oxygen evolution reaction (OER) is essential for energy storage and conversion applications, such as hydrogen generation from water splitting, rechargeable metal–air batteries and renewable fuel cells. Alkali metal cobalt phosphates show great potential as OER electrocatalysts. Herein, an original electrode design strategy is reported to realize an efficient OER electrocatalyst through engineering the coordination geometry of Co(II) in KCoPO4·H2O by a facile dehydration process. Experimental result indicated that the dehydration treatment is accompanied by a structural transformation from orthorhombic KCoPO4·H2O to hexagonal KCoPO4, involving a concomitant coordination geometry evolution of Co(II) from octahedral to tetrahedral configuration. More significantly, the local structural evolution leads to an advantageous electronic effect, i.e. increased Co–O covalency, resulting in an enhanced intrinsic OER activity. To be specific, the as-produced KCoPO4 can deliver a current density of 10 mA cm−2 at a low overpotential of 319 mV with a small Tafel slope of 61.8 mV dec−1 in alkaline electrolyte. Thus, this present research provides a new way of developing alkali metal transition-metal phosphates for efficient and stable electrocatalytic oxygen evolution reaction.


 Please wait while we load your content...
Please wait while we load your content...