The efficient degradation of organic pollutants in an aqueous environment under visible light irradiation by persulfate catalytically activated with kaolin-Fe2O3
Abstract
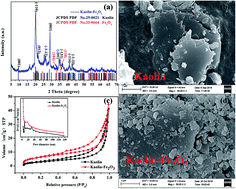
In recent years, persulfate (PS) has been widely studied as a promising oxidant. In this work, a new K-Fe2O3 catalyst was synthesized via a facile impregnation method. K-Fe2O3 samples were utilized as heterogeneous photocatalysts for the degradation of aquatic organic pollutants (rhodamine, RhB, and ciprofloxacin, CIP). The catalysts showed excellent catalytic activity in the presence of PS under the irradiation of visible light, owing to the generation of SO4˙− and ·OH active radicals. The degradation ratio and COD removal ratio for RhB were 99.8% and 88.3%. More importantly, the system retained a high degradation activity for RhB within a wide operating pH range of 2.9–10. The results of cycling degradation experiments confirmed that the K-Fe2O3 catalyst was stable and recoverable. Large-scale experiments for treating dye wastewater under irradiation by natural sunlight were carried out, showing that this study can provide a new perspective for the treatment of wastewater.


 Please wait while we load your content...
Please wait while we load your content...