Development and validation of a headspace needle-trap method for rapid quantitative estimation of butylated hydroxytoluene from cosmetics by hand-portable GC-MS
Abstract
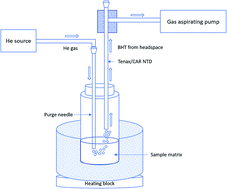
Butylated hydroxytoluene (BHT) is widely used as a stable and inexpensive antioxidant in skin care products. BHT is effective at penetrating the epidermis, and prolonged exposure to BHT has been reported to have toxic effects on the lungs, liver, and kidneys. Given that the carcinogenic factor of BHT is closely related to the dose and the length of exposure of the compound on a specific organ, quantitative estimation of BHT in BHT-containing cosmetics is very important in terms of quality control measurements for products used in daily life. In this study, we have developed a new method based on purge-and-trap technology using a headspace needle trap device (NTD) in addition to a comparative SPME methodology which was both coupled to portable gas chromatography (GC) toroidal ion trap mass spectrometer (MS) for rapid determination of BHT in cosmetics. The sample was placed in a glass vial and heated to 60 °C for 30 min to promote partitioning of BHT into the headspace, where it was then pre-concentrated by the Tenax/carboxen NTD. To verify these results, a divinylbenzene/polydimethylsiloxane SPME fiber was also used for headspace extraction. To quantify BHT in a sample, a standard addition calibration method was employed. A six-point calibration curve showed high linearity (R2 = 0.98) within a dynamic concentration range of 1000–10 000 ng mL−1 BHT in the sample matrix. Estimation of BHT by NTD yielded a concentration value of around 5000 ng/0.1 g body lotion, whereas the measurement of BHT using the SPME method yielded a value of around 5500 ng/0.1 g body lotion. The attained results indicate that the amount of BHT estimated by the proposed needle trap method was well-matched with the standard SPME technique. The results obtained from both methods were well matched (∼90%). One of the advantages imparted by this method is that it is based on headspace sampling instead of direct extraction of BHT via direct immersion into the sample matrix; as such, the method does not contaminate the instrument after sampling. Moreover, the use of a portable GC-MS enables on-site quality control measurements of healthcare products, thus circumventing the need for transportation and laborious laboratory sample preparation protocols.


 Please wait while we load your content...
Please wait while we load your content...