Recent advances in metal catalyzed or mediated cyclization/functionalization of alkynes to construct isoxazoles
Abstract
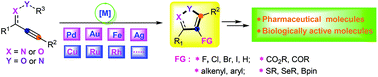
Isoxazoles and their derivatives represent privileged heteroaromatic motifs, which exhibit remarkable biological and pharmaceutical activities. Notably, metal catalyzed or mediated cyclization/functionalization of alkynes such as alkynone oxime ethers, acetylinic oximes, propargylic amino ethers, alkynyl nitrile oxides, and propargylic alcohols has recently attracted much attention from synthetic organic chemists because of their high efficiency in the assembly of structurally diverse isoxazole architectures under mild conditions with exceptional chemo-, stereo- and regioselectivities. In this review, a wide array of metal catalysts, such as palladium, copper, iron, gold, silver, and other metals, have been summarized and discussed in detail. Moreover, we also highlight some representative synthetic strategies along with reaction mechanisms involved during the reaction. And, the literature has been surveyed from 2010 until the beginning of 2020.


 Please wait while we load your content...
Please wait while we load your content...