Investigation of the remote acyl group participation in glycosylation from conformational perspectives by using trichloroacetimidate as the acetyl surrogate†
Abstract
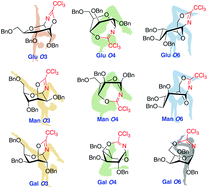
Unlike the O2 neighboring group participation in glycosylation, the participating effect of esters located on more remote positions of glycopyranosyl donors has not been unambiguously established and provokes plenty of questions. Here we systematically trapped the participation intermediates on three common pyranoses based on a ‘bis-trichloroacetimidates method’, which showed that trichloroacetimidate group participations from O4 and O6 are all quite possible except for D-mannopyranosyl O4. Besides, with the trapped participation intermediates in hand, the NMR data was collected to assist the DFT calculations to build up the most possible participating conformations, which, in turn, predicted the potentials of remote participation by trichloroacetimidates at different positions on the same donor. Finally, the ‘tris-trichloroacetimidates method’ was employed to support the computational calculations. This work provided a conformational perspective on understanding the potent remote acyl group participation in glycosylation reaction.


 Please wait while we load your content...
Please wait while we load your content...