Symmetrical and unsymmetrical fluorine-rich ullazines via controlled cycloaromatizations†
Abstract
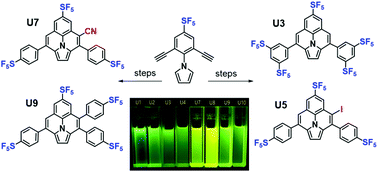
We report the synthesis of a series of fluorine-rich ullazines via well-controlled cycloaromatizations of highly electron-deficient alkynes. Specifically, three symmetrically-functionalized and three unsymmetrically-functionalized ullazine cores were constructed from 1-(2,6-dialkynylphenyl)-1H-pyrroles via controlled electrophilic cyclizations of highly electron-poor pentafluorosulfanylated (i.e., SF5-containing) phenylethynyl moieties onto the pyrrole. While the diminished reactivity of the electron-poor alkynes was initially problematic, this feature was subsequently found to be pivotal in facilitating thermal control of mono- versus double 6-endo-trig ring-closure in the key step. Iodinated ullazine products were shown to undergo further facile transformation into more complex unsymmetrical targets. The synthetic strategies reported herein grant access to a host of symmetrically- and unsymmetrically-decorated fluorine-rich ullazines with potential value as light-harvesting materials (e.g., dye-sensitized solar cells, organic photovoltaics) or as advanced intermediates for further synthetic elaboration.


 Please wait while we load your content...
Please wait while we load your content...