Understanding the lipid production mechanism in Euglena gracilis with a fast-response AIEgen bioprobe, DPAS†
Abstract
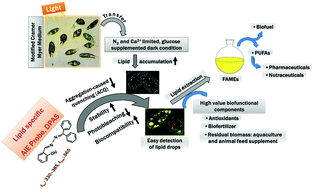
Lipid bodies are lipid-rich organelles that can regulate the storage of neutral lipids as energy sources in organisms. Visualisation of lipid droplets is an effective approach to understand lipid dynamics in microalgae. This study explores the required environmental conditions to yield lipid in a microalgal species Euglena gracilis as the biofunctional component using a lipid-specific aggregation-induced emission fluorogen, DPAS (C20H16N2O), and compares it to the commercial lipid staining probe BODIPY for visualising the lipid production in vivo. Five treatments are investigated for lipid production: (1) modified Cramer–Myers medium (MCM), (2) MCM without nitrogen (−), (3) MCM without nitrogen (−) and calcium (−), (4) MCM without nitrogen (−) and calcium (−), but with glucose (+), and (5) MCM without nitrogen (−) and calcium (−), but with glucose (+). Illumination was continuous at a rate of 70 mmol photons per m−2 s−1 in all treatments except with no light for treatment of 5. Distinct lipid droplets are labelled with DPAS and detected by confocal microscopy and flow cytometry to clarify the understanding of the lipid enrichment mechanism under various conditions. Treatment 1 indicates low lipid production in E. gracilis under an autotrophic condition. DPAS benefits from a very low background signal, and therefore, it is more sensitive than BODIPY for semiquantitative in vivo fluorescence measurements. Co-staining in the presence of BODIPY and chlorophyll also indicates that DPAS is suitable for multicolour imaging with red and green fluorophores. The present study demonstrates that DPAS is a highly effective biocompatible and photostable fluorophore for rapid and sensitive visualisation of lipid droplets. This novel staining method could be used to screen microalgae that have a potential to produce lipid droplets as a health supplement for humans.
- This article is part of the themed collections: FOCUS: Recent progress on aggregation-induced emission and Luminogenic bioprobes for personal health technologies


 Please wait while we load your content...
Please wait while we load your content...