Pyrazoloanthrone-functionalized fluorescent copolymer for the detection and rapid analysis of nitroaromatics†
Abstract
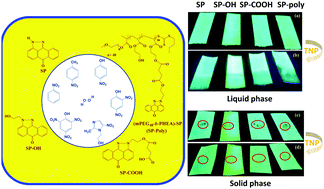
The development of sensors for rapid detection of chemical explosives with high sensitivity and selectivity is the focus of many research groups. In this work, we have developed a simple and straightforward synthesis of a block (co)polymer functionalized with pyrazoloanthrone (SP). The block (co)polymer was synthesized via reversible addition–fragmentation chain-transfer (RAFT) polymerization of 2-hydroxyethyl acrylate using a PEG-functionalized RAFT-agent. Subsequently, the SP was coupled to the poly(2-hydroxyethyl acrylate) block through DCC coupling. The structural and physicochemical properties of the (co)polymer were studied to explore its potential towards the detection of nitroaromatics as a model for explosives. A systematic comparison is made on the chemosensing behavior of the (co)polymer and three small molecule pyrazoloanthrone analogues with different functional groups (SP, SP–OH and SP–COOH). Fluorescence studies demonstrated a significant decrease in the fluorescence intensity of the four fluorophores in the presence of different nitroaromatics and showed unprecedented selectivity for 2,4,6 trinitrophenol (TNP). The Stern–Volmer rate constants (Ksv) of the SP-functionalized copolymer (Ksv = 9.74 × 104 M−1) showed an ∼3.7 times higher quenching rate constant than its monomer analog (SP) for TNP with a limit of detection (LOD) of 19 ppm. A static quenching mechanism with photoinduced electron transfer process, intermolecular hydrogen bonding and electrostatic interactions induce turn off fluorescence behavior. The interference studies with other nitroaromatics in an aqueous medium and real-time analysis in the solid-state methods demonstrate the potential of the block (co)polymer towards practical applications.
- This article is part of the themed collection: 2020 Materials Chemistry Frontiers HOT articles


 Please wait while we load your content...
Please wait while we load your content...