Small organic molecule-based nanoparticles with red/near-infrared aggregation-induced emission for bioimaging and PDT/PTT synergistic therapy†
Abstract
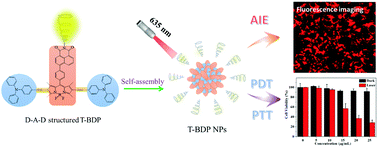
Organic nanomaterials with efficient ROS generation and high photothermal conversion efficiency have emerged as a new generation therapeutic agent for PDT/PTT synergistic cancer treatment. However, most of the organic nanomaterials exhibited weak emission in water because of the aggregation-caused quenching (ACQ) effect, which seriously hampers their biological application in fluorescence bioimaging. In this study, we design and synthesize a new organic small molecule (T-BDP) with a donor–acceptor–donor (D–A–D) structure by the conjugation of boron-dipyrromethene (BODIPY) and triphenylamine (TPA). Moreover, a 1,8-naphthalenediimide (NI) structure with electron-withdrawing ability is introduced onto the core of BODIPY to further enhance the intramolecular charge transfer, leading to the redshift of absorption to the near-infrared region. T-BDP displays significant aggregation-induced emission (AIE) performance, probably due to the presence of two twisted TPA groups onto the BODIPY core. Accordingly, after self-assembly into nanoparticles, T-BDP NPs exhibit strong near-infrared emission in water. Under single 635 nm laser irradiation, T-BDP NPs could generate reactive oxygen species and heat simultaneously. The photothermal conversion efficiency of T-BDP NPs is determined to be 50.9%. The low dark toxicity and high photocytotoxicity of T-BDP NPs are verified against A549 cells using the MTT assay and the AM/PI staining method. Due to the strong emission of T-BDP NPs, their accumulation and subcellular localization in cancer cells are observed using a laser confocal fluorescence microscope. The results demonstrate that T-BDP NPs are mainly located in the lysosomes of cancer cells. Thus, the as-prepared small molecule-based AIE nanoparticles hold great potential for fluorescence imaging-guided PDT/PTT synergistic tumor therapy.
- This article is part of the themed collections: FOCUS: Recent progress on bioimaging technologies and FOCUS: Recent progress on aggregation-induced emission


 Please wait while we load your content...
Please wait while we load your content...