Tuning singlet fission in amphipathic tetracene nanoparticles by controlling the molecular packing with side-group engineering†
Abstract
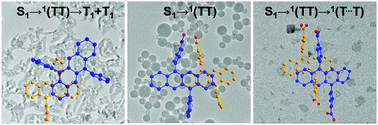
Three tetracene derivatives with one (DPhTc-COOH), two (DPhTc-(COOH)2) or no (DPhTc) carboxylic groups are synthesized and prepared into colloidal nanoparticles in aqueous solution. Theoretical simulation shows that various molecular arrangements are adopted in these three nanoparticles. So different electronic coupling strengths between tetracene units are achieved in these three nanoparticles. Transient absorption spectra demonstrate that SF can occur in all nanoparticles but with different SF dynamics and efficiencies depending on the coupling strength among the adjacent tetracene units. In DPhTc-COOH NPs with strong interactions, efficient SF can proceed quickly, but the resulting triplet pair cannot dissociate into free triplets and recombines into the ground state quickly. When the interaction decreases in DPhTc-(COOH)2 and DPhTc NPs, SF will become slow and less efficient but the triplet pair can dissociate into free triplets or partially separated triplets. Our results demonstrate successfully that the number of carboxylic groups influences the tetracene packing and coupling in nanoparticles, as well as the SF kinetics and efficiency. Strong interactions between tetracene units are necessary for quick and efficient SF, but the formed triplet pair cannot separate into a long-lived triplet state. To get a high free triplet yield, elaborate tuning of the interaction among the tetracene molecules to achieve quick dissociation of the triplet pair is necessary.


 Please wait while we load your content...
Please wait while we load your content...