NH4V3O8·0.5H2O nanobelts with intercalated water molecules as a high performance zinc ion battery cathode†
Abstract
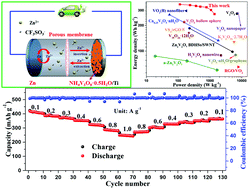
Aqueous rechargeable Zn-ion batteries (ARZIBs) have been attracting huge attention recently, where V-based materials with host layer structures and fast channels enable the efficient diffusion of metal ions, leading to excellent properties of Zn2+ storage. Several ammonium vanadates have been explored as potential cathodes, and their performance in ARZIBs varies considerably. Herein, we choose H2O-intercalated NH4V3O8 (NH4V3O8·0.5H2O) nanobelts, which are synthesized by a low-temperature hydrothermal process, and reveal that the electrochemical performance of NH4V3O8 is strongly enhanced by the H2O molecules intercalated in the layer structure. Indeed, the NH4V3O8·0.5H2O nanobelts exhibit a super-high capacity of 423 mA h g−1 at 0.1 A g−1, together with long-term stability (50.1% retention after 1000 cycles) at 1 A g−1. The Zn//NH4V3O8·0.5H2O battery thus assembled delivers a high energy density of 353 W h kg−1 at a power density of 114 W kg−1, comparing favorably with most of the state-of-the-art V-based cathode materials reported for ARZIBs. As a promising cathode candidate for aqueous batteries, the reversible (de)intercalation of Zn2+ in the H2O-intercalated NH4V3O8·0.5H2O gives rise to the formation of Zn3(OH)2V2O7·2H2O, which helps retain the desired long-term stability.
- This article is part of the themed collection: Battery science and technology – powered by chemistry


 Please wait while we load your content...
Please wait while we load your content...