Near-infrared electron acceptors with fused nonacyclic molecular backbones for nonfullerene organic solar cells†
Abstract
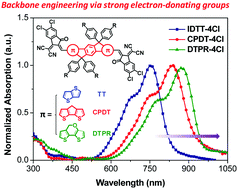
Core engineering of small molecule acceptors (SMAs) is crucially important for enhancing device efficiency for nonfullerene organic solar cells (NF-OSCs). The most commonly used SMAs (e.g., ITIC) utilize indacenodithieno[3,2-b]thiophene (IDTT) as the central core, which has restricted their absorption ranges due to the weak electron-donating ability and short conjugation length. Here, we fused two electron-rich units, namely cyclopenta[2,1-b:3,4-b′]dithiophene (CPDT) and dithieno[3,2-b:2′,3′-d]pyran (DTPR), into the cores for constructing low-bandgap SMAs. The resulting CPDT-4Cl and DTPR-4Cl molecules exhibit extended nonacyclic central cores and strengthened intramolecular transfer (ICT) effect, resulting in red-shifted absorption (up to ∼950 nm) and up-shifted HOMO levels compared with IDTT-4Cl. Consequently, the NF-OSCs based on PTB7-Th:CPDT-4Cl and PTB7-Th:DTPR-4Cl achieved higher PCEs of 12.15% and 10.75%, respectively, than those of the PTB7-Th:IDTT-4Cl ones (7.70%). Notably, high short-circuit current densities (JSC) of 23–25 mA cm−2 were obtained by the CPDT-4Cl and DTPR-4Cl-based devices, indicating the great potential of the electron-donating CPDT and DTPR as promising building blocks to construct high-performance low-bandgap SMAs.


 Please wait while we load your content...
Please wait while we load your content...
