In situ formed methyl-co-(bis-R) silsesquioxane based polymer networks with solvent controlled pore size distributions and high surface areas†
Abstract
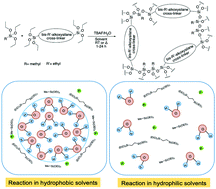
High surface area materials are of considerable interest for gas and analyte storage/capture, molecular sieving, catalyst supports, and slow release delivery systems. This paper set out to gain a fundamental understanding of the influence reaction parameters have on structure–property relationships of fluoride-catalyzed methylsilsesquioxane based porous networks. For this study, twelve solvents of varying polarities were used and were compared for their effect on gelation and final properties related to porosity, hydrophobicity, and solvent uptake. We find that solvent choice and water compatibility plays an extremely important role in gaining specific pore size distributions, surface areas and gel types, with a consistent cross-linker size and length. Reactions conducted in acetonitrile and dichloromethane offer surface areas >1000 m2 g−1, but with acetonitrile being mesopore dominant and having global gel formation versus dichloromethane, which is micropore dominant and gave gel particles. The difference in these reactions comes from their variances in solvation of monomer/oligomer and water. In addition to these findings, swelling studies of gels made from acetonitrile result in >700% by mass solvent reabsorption after drying out. This study enhances the toolbox for continued improvement and development of thermally stable, selective analyte uptake, porous materials.
- This article is part of the themed collection: 2020 Materials Chemistry Frontiers HOT articles


 Please wait while we load your content...
Please wait while we load your content...