Understanding of copolymers containing pyridine and selenophene simultaneously and their polarity conversion in transistors†
Abstract
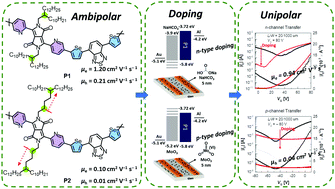
By simultaneously considering the powerful electron-accepting capability of pyridine and the strong selenophene (Se)–Se interaction of selenophene, we present here the design and synthesis of both pyridine and selenophene-containing, pyridine-flanked diketopyrrolopyrrole and 4,7-di(selenophen-2-yl)-2,1,3-benzothiadiazole-based copolymers (P1 and P2) for use in organic field-effect transistors, where P1 has a shorter branched position of the side chains in relation to the backbone as compared to P2. The structure–property relationships associated with branching point engineering in the copolymers are established by applying a range of technical analyses. Overall, P1 interestingly exhibits both a higher hole and electron mobility of up to 1.2 cm2 V−1 s−1 and 0.21 cm2 V−1 s−1, respectively, which are over one order of magnitude higher than that of P2. This observation demonstrates the opposite view of the branching points away from the backbone for high-performing organic field-effect transistors. Besides, by doping sodium bicarbonate and molybdenum trioxide with electrodes, we succeeded in affecting the transition from ambipolarity of P1 to unipolar n- and p-type characteristics, respectively. These findings improve our understanding of both the role of branching point engineering in enhancing the carrier transport and the use of doping for switching the polarity of P1.
- This article is part of the themed collection: Celebrating Prof. Fred Wudl’s 80th Birthday


 Please wait while we load your content...
Please wait while we load your content...