The origin of the unusual red-shifted aggregation-state emission of triphenylamine-imidazole molecules: excimers or a photochemical reaction?†
Abstract
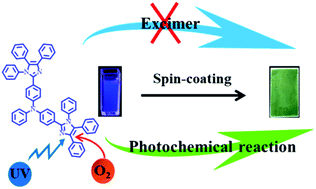
At the moment, there is still no consensus regarding the origin of the newly apparent unusual red-shifted aggregation-state emission arising from some triphenylamine-imidazole luminescent materials. One view is that the unusual emission originates from the formation of excimers, but a recently observed photochemical reaction also seems to contribute to it. Herein, a new imidazole molecule, TPDBM, was synthesized to investigate the actual origin of this phenomenon. Compared with a dilute solution of TPDBM, its film obviously exhibits newly apparent high red-shifted emission at around 520 nm in the PL spectrum. According to UV-vis spectra and aggregation-induced emission (AIE) and concentration-dependent emission results, it seems that the red-shifted emission is derived from excimers. However, further studies of excitation spectra, in situ PL spectra and oxygen-free PL spectra entirely overturn this conclusion and ultimately confirm that it seems to come from a photochemical reaction occurring on the imidazole ring of TPDBM. Two photochemical products, TPBMZ and TPDYZ, with excellent AIE effects were obtained through this photochemical reaction, and TPBMZ can be further oxidized to TPDYZ due to the quite fast reaction rate. Furthermore, the photochemical reaction of TPDBM doped into PMMA film was suitably applied as a fluorescent probe for oxygen with a lowest detectable oxygen volume ratio of 0.1%.
- This article is part of the themed collection: Recent Progress on Aggregation-Induced Emission


 Please wait while we load your content...
Please wait while we load your content...