In situ hydroxyl radical generation using the synergism of the Co–Ni bimetallic centres of a developed nanocatalyst with potent efficiency for degrading toxic water pollutants†
Abstract
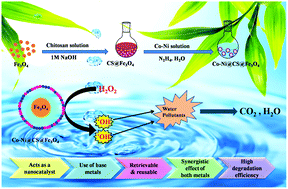
In today's global society, the development of novel materials for wastewater treatment has attracted the interest of academic and industrial researchers as this can ensure reliable access to clean water. Herein, we report for the first time the development of an economical and sustainable bimetallic nanocatalytic system comprising a polymeric magnetic chitosan support decorated with redox couple Co–Ni nanoparticles and highlight its role in the in situ generation of hydroxyl radical species that can effectively catalyze the degradation of toxic water contaminants, including the pesticide 2,4-dichlorophenoxyacetic acid (2,4-D) and the azo dye methyl orange (MO). Coumarin fluorescence probe and radical scavenging studies provided valuable insights into the mechanistic pathway, revealing the in situ generation of hydroxyl radicals that were responsible for expediting the degradation process. Kinetics studies revealed that the degradation process followed pseudo-first-order kinetics in both cases (with kinetic constant values of 0.07517 min−1 for 2,4-D and 0.03352 min−1 for MO at room temperature under neutral pH conditions). In addition, reusability studies suggested that the Co–Ni@CS@Fe3O4 nanocatalyst maintained its degradation efficacy even after eight consecutive cycles. Notably, this is the first report that effectively exploits a synergistic combination of two transition metals, Co and Ni, anchored onto the surface of chitosan-coated Fe3O4 nanoparticles to effectively catalyze the degradation of hazardous contaminants in the presence of a green oxidant under neutral and ambient conditions.


 Please wait while we load your content...
Please wait while we load your content...