Aggregation-induced emission characteristics of o-carborane-functionalized fluorene and its heteroanalogs: the influence of heteroatoms on photoluminescence†
Abstract
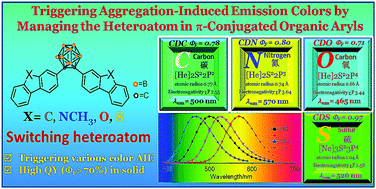
o-Carborane was used to alter the electronic states of π-conjugated organic aryls and was demonstrated as an effective optical and electronic control unit to tune the photophysical properties, endowing the molecules with certain luminescence properties and modifying the HOMO and LUMO energies for electron transfer. We introduced fluorene and its heteroanalogs (carbazole, dibenzofuran and dibenzothiophene), four types of substitutes with different electron-rich or electron-deficient heteroatoms, to the two carbon atoms of o-carborane to synthesize CDC, CDN, CDO and CDSvia a modified nickel-catalyzed cross-coupling reaction. The photophysical properties of o-carborane-containing heterocyclic compounds were investigated and compared with each other. The results showed that all compounds were AIE luminogens with tunable fluorescence colors from blue to yellow emissions (λem = 465–570 nm) and possessed excellent emission quantum efficiencies in the solid state (ΦF > 70%). Among the four o-carborane-containing heterocyclic compounds, the ground state structures confirmed by crystallographic measurements provided a close correlation of the structure and electronic properties between the π-conjugated organic aryl groups and o-carborane. Besides, the heteroatoms controlled and altered the electronic interaction between aryls and o-carborane due to the different electron-rich or electron-deficient attributes. The HOMO and LUMO energies were estimated by DFT calculations. The excited states were calculated by TD-DFT and correlated with the corresponding absorption and emission spectra to systematically elucidate the electronic perturbation. This work not only demonstrates an efficient strategy for constructing o-carborane-containing heterocyclic AIE luminogens but also indicates that they are promising as advanced optoelectronic materials.
- This article is part of the themed collection: Recent Progress on Aggregation-Induced Emission


 Please wait while we load your content...
Please wait while we load your content...