Dithienothiazine dimers, trimers and polymers – novel electron-rich donors with red-shifted luminescence†‡
Abstract
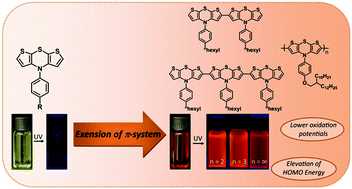
Starting from N-aryldithienothiazine, a tool box of various iodinated and borylated dithienothiazines was generated by a straightforward α-lithiation and electrophilic trapping of thiophene cores. By using lithiation–borylation-Suzuki (LiBS) and lithiation–zincation-Negishi (LiZiN) sequences π-conjugated dithienothiazine oligo- and polymers were synthesized in a one-pot fashion and in moderate to good yields. Studies on the photophysical and electrochemical properties reveal that an increasing number of dithienothiazine units causes a bathochromic shift in the absorption and orange to red fluorescence in solution, as quantified by absorption and emission spectroscopy and assigned by DFT calculations. Furthermore, from cyclic voltammetry measurements an elevation of the HOMO energy level with increasing number of dithienothiazine units and concomitantly diminishing optical bandgaps were determined, suggesting that the title compounds are suitable as novel donor systems for organic hole transport materials.


 Please wait while we load your content...
Please wait while we load your content...