Palladium bis-pincer complexes with controlled rigidity and inter-metal distance†
Abstract
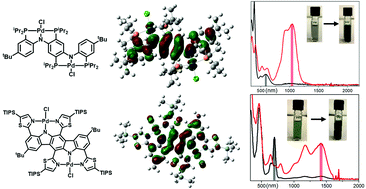
We report a series of redox-active bis(pincer) Pd(II) complexes in which the redox active units are based on either a diarylamido or a carbazolide framework. Compounds 1 and 2 contain two full diarylamido/bis(pincer) PNP units connected either via an Ar–O–Ar linker (1) or an Ar–Ar bond (2). Compound 3 is a fused bis(pincer) where the two PNP units share an aromatic ring. Compound 4 is built around an indolo[3,2-b]carbazole core in which two NNN pincers share an aromatic ring similarly to 3. These metal complexes all display two reversible oxidation waves with the ΔE values increasing in the order of 1 < 2 < 4 < 3. The same trend in increasing electronic coupling emerges from the analysis of the IV-CT bands in the NIR portion of the optical spectra. The analysis of these compounds was further advanced by data from EPR spectroscopy, X-ray diffractometry, and DFT calculations. It is concluded that the monooxidized cations 2+–4+ belong to Class III on the Robin-Day classification of mixed-valence compounds. Compound 4 possesses enforced near-planarity that enables delocalization of the unpaired electron in 4+ across a broader conjugated system compared to 3+.


 Please wait while we load your content...
Please wait while we load your content...