Nitric oxide dioxygenation (NOD) reactions of CoIII-peroxo and NiIII-peroxo complexes: NOD versus NO activation†
Abstract
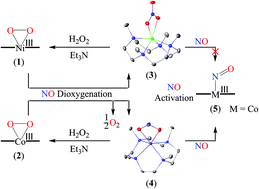
Here, we report the nitric oxide dioxygenation (NOD) reactions of NiIII-peroxo, [(12-TMC)NiIII(O22−)]+ (NiIII–O22−, 1), and CoIII-peroxo, [(12-TMC)CoIII(O22−)]+ (CoIII–O22−, 2), complexes bearing N-tetramethylated cyclam (TMC) ligands. The reaction of complex 1 with nitric oxide (NO) generates NiII-nitrato complex [(12-TMC)NiII(NO3−)]+ (NiII-NO3−, 3). However, in contrast to the NOD reaction of 1, compound 2 produces CoII-nitrito complex, [(12-TMC)CoII(NO2−)]+ (CoII-NO2−, 4), with O2 gas formation. Results suggested the formation of a presumptive Ni-peroxynitrite ([Ni-PN]+, [Ni-ONOO–]+) intermediate, in the NOD reaction of 1, prior to the generation of NiII-NO3− (3). Although the end product of the reaction of 2 with NO is different from that of 1, the mechanistic study explored by using 2,4-di-tert-butylphenol (2,4-DTBP) suggested that the reaction of 2 with NO also occurs via a presumed Co-peroxynitrite ([Co-PN]+, [Co-ONOO−]+) intermediate. Furthermore, exploring these NOD reactions and tracking the oxygen atom using 18O-labeled hydrogen peroxide (H218O2) revealed that two oxygen atoms of 3 (NiII-18O2N16O−) and one oxygen atom of 4 (CoII-18ON16O−) are derived from the peroxo (O22−) moieties of 1 and 2, respectively. Furthermore, we have explored the M-peroxo regeneration from NOD products (3 and 4), and for the first time, we have observed the formation of MIII–O22− complexes, 1 and 2, upon treatment with H2O2 and triethylamine (TEA), respectively. This work is the first-ever report which showed the exact NO dioxygenase (NOD) enzyme catalytic cycle. Furthermore, we have also explored the NO activation of 3 and 4, and surprisingly, we observed the formation of Co-nitrosyl (5) from 4 upon reaction with NO; in contrast, 3 was unreactive towards NO. Evidence supporting the formation of [Ni-PN]+ and [Co-PN]+ intermediates is also presented.


 Please wait while we load your content...
Please wait while we load your content...