Surface defect engineering of mesoporous Cu/ZnS nanocrystal-linked networks for improved visible-light photocatalytic hydrogen production†
Abstract
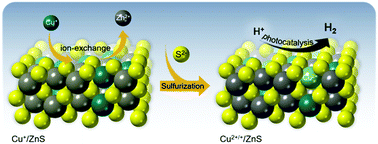
Transition metal sulfides have been emerging as one of the most attractive and prospective catalysts for the direct conversion of solar energy into chemical fuels. Their intriguing compositional and electronic characteristics and their feasibility for integration in porous architectures endow metal sulfide materials with superior activity for photochemical catalysis. In the present work, high-surface-area Cu-doped ZnS nanocrystal (NC)-linked mesoporous frameworks are successfully synthesized for use as cost-effective catalysts for photochemical hydrogen evolution. Benefiting from the suitable band-edge alignment and enhanced visible light absorption resulting from the interfacial charge transfer between ZnS and Cu2S NCs, there is a spatial separation of charge carriers which leads to excellent activity for photocatalytic hydrogen production. Moreover, the results obtained here show that surface defect passivation through a wet-chemical sulfidation process effectively increases the photochemical performance of the composite catalysts by improving the transport efficiency of electrons at the Cu2S/ZnS interface and changing the Helmholtz layer potential drop at the ZnS/Cu2S/electrolyte junction. Thus, a remarkable improvement of 1 mmol h−1 gcat−1 for hydrogen evolution is observed with the sulfide-treated Cu2S/ZnS catalyst containing 5 mol% Cu, which is associated with a 17.6% apparent quantum yield under 410 nm irradiation. This work provides an interesting strategy for enhancing the interface charge transfer properties and hydrogen evolution activity of metal sulfides by surface defect engineering with sulfide ions.
- This article is part of the themed collection: Recent Open Access Articles in Frontiers Journals


 Please wait while we load your content...
Please wait while we load your content...