Constructing a CeO2−x@CoFe-layered double hydroxide heterostructure as an improved electrocatalyst for highly efficient water oxidation†
Abstract
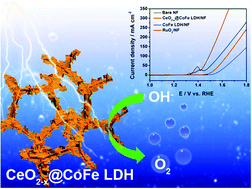
The oxygen evolution reaction (OER) plays an essential role in energy conversion, especially during the process of hydrogen production via electrocatalytic water-splitting technology. Herein, a heterostructure strategy to construct a CeO2−x decorated CoFe layer double hydroxide (LDH) grown on Ni foam (NF) (denoted as CeO2−x@CoFe LDH/NF) is presented. By constructing the CeO2−x@CoFe LDH heterostructure on NF, the as-prepared CeO2−x@CoFe LDH/NF offers superior electrocatalytic performance for the OER. CoFe LDH as a recognized catalyst provides active sites for OER reactions and CeO2−x nanoparticles bring more oxygen vacancies and synergies with the LDH structure to improve the catalytic performance of the OER. In 1.0 M KOH, it needs an ultralow overpotential of 204 mV to achieve the current density of 100 mA cm−2, and also shows long term durability for more than 30 hours. Moreover, when subjected to simulated alkaline seawater (1.0 M KOH + 0.5 M NaCl), CeO2−x@CoFe LDH/NF still maintains the high activity and durability for the OER without being affected by chloride species. This work will provide a guideline for designing heterostructures of nanoparticles and LDH to drive efficient water oxidation reactions in the future.


 Please wait while we load your content...
Please wait while we load your content...