The hexagonal perovskite Ba0.5Sr0.5Co0.8Fe0.2O3−δ as an efficient electrocatalyst for the oxygen evolution reaction†
Abstract
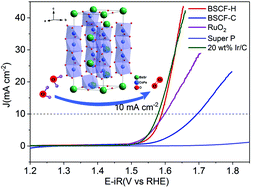
Oxygen evolution reaction (OER) electrocatalysts with high efficiency and durability are urgently needed for energy conversion and storage of clean energy. Herein, we report the finding in the development of a hexagonal Ba0.5Sr0.5Co0.8Fe0.2O3−δ (BSCF-H) perovskite as an OER electrocatalyst. Prepared by a sol–gel method, the BSCF-H perovskite displays excellent OER efficiency and stability in a harsh alkaline solution. The overpotential of BSCF-H reveals 360 mV at a current density of 10 mA cm−2 in 0.1 M KOH solution, and its catalytic activity for the OER is even comparable to that of the commercial Ir/C (20%) catalyst. Furthermore, BSCF-H possesses more operational stability with slight current reduction than cubic Ba0.5Sr0.5Co0.8Fe0.2O3−δ (BSCF-C) and RuO2 catalysts. This high activity is explained by a faster charge transfer rate, higher electrochemical surface area and a larger amount of oxygen species (O22−/O−). The Co d-band center and O p-band center of BSCF-H move to the Fermi level, which indicates the better OER performance of BSCF-H. This study demonstrates the new type of hexagonal Co-based perovskite catalyst that could be a low-cost alternative for electrochemical energy technology.


 Please wait while we load your content...
Please wait while we load your content...