Combined DFT and geometrical–topological analysis of Li-ion conductivity in complex hydrides†
Abstract
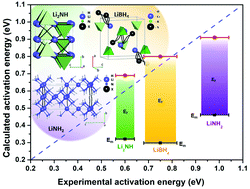
On the basis of DFT calculations, Li-ion migration was analyzed for LiBH4, LiNH2, Li2NH, Li2BH4NH2, Li4BH4(NH2)3 and Li5(BH4)3NH complex hydrides by means of the nudged elastic band method. In addition, a Voronoi-partition-based method, as implemented in the ToposPro program package, was adopted to determine cavities and channels in the complex hydrides and possible Li-ion migration pathways were computed. Experimental data for the Li-ion conductivity in the six compounds, measured by electrochemical impedance spectroscopy, have been taken from the literature and activation energies have been determined by a statistical analysis. A link between experimental and calculated activation energies has been evidenced, suggesting that topological analysis can provide good hints for the estimation of ion conductivity in complex hydrides.


 Please wait while we load your content...
Please wait while we load your content...