Template-directed assembly of urchin-like CoSx/Co-MOF as an efficient bifunctional electrocatalyst for overall water and urea electrolysis†
Abstract
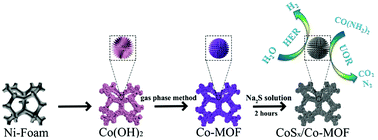
Since the theoretical potential of urea electrolysis is lower than that of water splitting, urea electrolysis is an ideal method for hydrogen production. In this work, we have synthesized an urchin-like Co-based metal–organic framework (Co-MOF) grown on a Co(OH)2 template by the gas phase method. As the specific surface area increased and more active sites were exposedcd, the low activity of the bulk MOF for electrocatalytic water splitting was overcome. Through a vulcanization reaction, surface vulcanized Co-MOF (CoSx/Co-MOF) was synthesized. CoSx/Co-MOF as a bifunctional catalyst has good catalytic performance toward the hydrogen evolution reaction (HER), oxygen evolution reaction (OER) and urea oxidation reaction (UOR). Compared with the catalytic activity of the OER, the potential of the UOR only needs 1.315 V (vs. RHE), while the potential of the OER needs 1.51 V (vs. RHE) at 10 mA cm−2. The catalytic activity in the urea electrolysis of a CoSx/Co-MOF||CoSx/Co-MOF electrolyzer is much better than that in an alkaline electrolyte. The CoSx/Co-MOF||CoSx/Co-MOF electrolyzer only needs a cell voltage of 1.48 V to achieve a current density of 10 mA cm−2 for a urea electrolytic cell in 1.0 M KOH with 0.5 M urea.


 Please wait while we load your content...
Please wait while we load your content...