Mechanism of oxygen vacancy assisted water-splitting of LaMnO3: inorganic perovskite prediction for fast solar thermochemical H2 production
Abstract
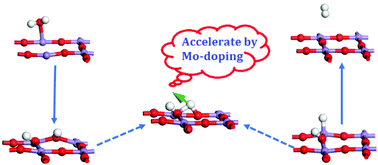
Highly reducible and thermally stable lanthanum–manganese perovskites represent a promising class of materials for solar thermochemical hydrogen production. However, such materials suffer from a low hydrogen production rate, resulting in an incomplete re-oxidation yield. To enhance the kinetic performance, the mechanism of water-splitting and hydrogen production around the oxygen vacancy of the LaMnO3(010) defective surface is revealed. A transition state with the activation energy Ea = 126.45 kJ mol−1 is explored in the rate-determining step of hydrogen transfer from superficial O to neighbor Mn. Based on the analysis of the Ea results, the H2 production time ratio between La0.9Sr0.4MnO3 and La0.9Sr0.1MnO3 is 47.3, close to the experimental data from the literature. Also, the kinetic unfavourability of Sr and the favorability of Al and Ga are predicted, which are in good agreement with previous experimental phenomena. Moreover, La0.875Sr0.125Mn0.875Mo0.125O3 is suggested as a promising material to rapidly produce H2. All the results demonstrate the effectiveness of the proposed water-splitting mechanism and provide an easy access for fast determination of kinetically favorable dopants for lanthanum–strontium–manganese perovskites.


 Please wait while we load your content...
Please wait while we load your content...