Rational design of Co-S-P nanosheet arrays as bifunctional electrocatalysts for both ethanol oxidation reaction and hydrogen evolution reaction†
Abstract
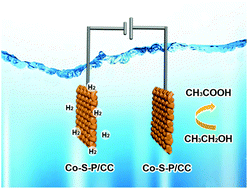
Highly active, ultra-long duration and cost-effective catalysts are imminently required for the development of electrolytic appliances for H2 generation. Herein, we propose a novel and facile strategy to fabricate P doped CoS2 nanosheet arrays on carbon cloth (Co-S-P/CC) as bifunctional electrocatalysts toward hybrid water electrolysis, in which the tardy anodic oxygen evolution reaction (OER) is substituted by a more favorable kinetic and thermodynamic ethanol oxidation reaction (EOR). Benefitting from the abundant active sites of Co-S-P nanosheets and the robust adhesion between Co-S-P and CC, the binder-free self-supported bifunctional Co-S-P/CC electrodes have satisfactory electrocatalytic activities and stabilities toward the EOR and hydrogen evolution reaction (HER). Notably, owing to the more favorable kinetics and thermodynamics of the EOR compared to those of the OER catalyzed by the Co-S-P/CC electrode, the required cell voltage for hybrid water electrolysis (1.63 V) is remarkably decreased compared to water electrolysis (1.77 V). Furthermore, the oxidation product acetic acid is more valuable than raw ethanol or oxygen from water splitting alone. This work may broaden the horizons in exploring and designing novel electrocatalysts, which can integrate the HER with other small organic molecule oxidation reactions for multifarious energy-related applications.


 Please wait while we load your content...
Please wait while we load your content...