Slow magnetic relaxation in hexacoordinated cobalt(ii) field-induced single-ion magnets†
Abstract
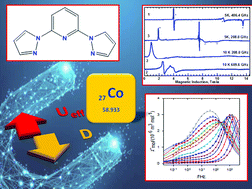
To gain a better insight into the factors affecting the enhancement of the energy barrier in single molecule (single ion) magnets, three hexacoordinate cobalt(II) complexes based on the tridentate ligand 2,6-bis(pyrazol-1-yl)pyridine (pypz) and pseudohalide ions have been synthesized and investigated. It was found that dicyanoamido and azido ligands act as bridges to form a one dimensional network based on a single μ1,5-dca bridge [Co(pypz)(dca)(H2O]·dca (1) and dimer [Co2(pypz)2(μ1,1-N3)2(N3)2]·2CH3OH (2), while tcm− counterbalanced the charge of [Co(pypz)2]2+ in [Co(pypz)2](tcm)2 (3), where dca− = dicyanamide ion; tcm− = tricyanomethanide ion, respectively. The DC magnetic data show a sizable magnetic anisotropy, which was confirmed by high-field/high frequency EPR measurements. Two of them are SIMs (1 and 3) and the other one is a SMM (2). All complexes exhibit field induced slow magnetic relaxation with two (1 and 2) or three relaxation channels (3) and an exceptionally long relaxation time for the low-frequency channel upon application of an external field BDC = 0.4, 0.3, and 0.4 T at T = 1.9 K; τ(LF) = 1.9, 2.1 and 0.9 s, respectively. Additionally, the high spin-reversal barriers, U = 103 K for 1 and 95 K for 2, are among the largest for field-induced SIMs for cobalt(II) reported in the literature.


 Please wait while we load your content...
Please wait while we load your content...