Unraveling the complexation mechanism of actinide(iii) and lanthanide(iii) with a new tetradentate phenanthroline-derived phosphonate ligand†
Abstract
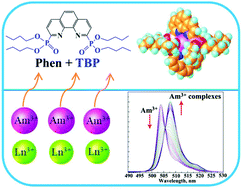
The extraction and complexation of trivalent Am(III) and lanthanide(III) with a new phenanthroline-derived phosphonate ligand, tetrabutyl-(1,10-phenanthrolin-2,9-diyl)phosphonate (C4-POPhen), were investigated by solvent extraction, absorption spectroscopy, NMR titration, luminescence spectroscopy, and theoretical computation. Selective extraction of Am(III) over Eu(III) from highly acidic HNO3 solution (up to 3.0 M) with good selectivity and a fast extraction rate was achieved by using the C4-POPhen ligand. The 1 : 2 metal/ligand complex was found to be the predominant species in the extracted organic phase by a slope analysis method. NMR titration results demonstrated that only a 1 : 1 complex of C4-POPhen with Ln(NO3)3 was formed in a CH3OH system, while two complexes (1 : 1 and 1 : 2) were found when using Ln(ClO4)3 as the metal salts instead of Ln(NO3)3. The stability constants of the C4-POPhen/Am(III) and C4-POPhen/Ln(III) complexes in CH3OH with perchlorate and nitrate were determined by absorption spectroscopy. The Am(III) complexes exhibited higher complexation ability than Ln(III), which was consistent with the extraction results. DFT calculations also suggested that the Am–N bonds in C4-POPhen/Am(III) complexes had a higher degree of covalence than the Eu–N bonds in C4-POPhen/Eu(III) complexes, revealing the underlying driving force for the extraction selectivity of Am(III) over Eu(III) by C4-POPhen. These results indicate that phenanthroline-derived phosphate ligands may serve as promising candidates for separation of trivalent actinides over lanthanides from highly active nuclear waste.


 Please wait while we load your content...
Please wait while we load your content...