Metal–organic framework derived nitrogen-doped carbon-RhNi alloys anchored on graphene for highly efficient hydrogen evolution reaction†
Abstract
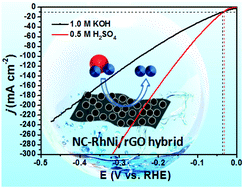
Synthesizing cost-effective, highly active and stable electrocatalysts for hydrogen evolution reaction (HER) is incredibly critical for scalable hydrogen production through electrochemical water splitting, yet it remains a significant challenge. Noble metal-based materials are generally considered the most excellent electrocatalysts for HER, but their scarcity and high cost impede their comprehensive application. Herein, an auspicious strategy to couple highly active precious metals with transition metals as excellent HER catalysts is presented, which not only can effectively reduce the dosage of the precious metals, but also greatly improve the performance of catalysts. The desired electrocatalysts, based on nitrogen-doped carbon-RhNi alloy nanoparticles anchored on reduced graphene oxide nanosheets (NC-RhNi/rGO), are obtained from pyrolysis of bimetallic RhNi-MOFs/GO precursor. The as-prepared NC-RhNi/rGO catalyst achieves noticeable HER performance, with overpotentials of 37 and 34 mV at the current density of 10 mA cm−2 in 1.0 M KOH and 0.5 M H2SO4, respectively, which are better than that of commercial Pt/C. Experimental results reveal that the superior activities are a benefit of the synergistic effect of bimetallic alloy and the collective effects of unique architecture, where the synergistic effect can facilitate electron transfer and the unique architecture provides rich active sites. This work provides a promising approach for the synthesis of highly efficient nanocomposite electrocatalysts for HER.


 Please wait while we load your content...
Please wait while we load your content...