Nanofabrication of Au nanoparticles over conductive metallohydrogel nanofibers for nanocatalysis application†
Abstract
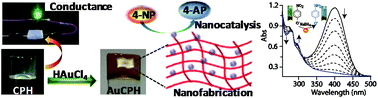
A conductive and fluorescent metallohydrogel (1% w/v, CPH) involving coordination polymers has been synthesized from non-fluorescent components viz., an L-tyrosine derived pro-ligand (1), LiOH and Zn(NO3)2 used in 1 : 3 : 1 proportion at pH 11–12. CPH shows reversible gel–sol transitions in response to external stimuli including thermal, mechanical and ultrasound stress. CPH also exhibits good shape persistence and reswelling properties. The formation of a true gel phase was established on the ground of detailed rheological experiments. Each step involved in the formation of CPH, including initial coordination, polymerization, aggregation, nanofiber growth followed by gelation, has been thoroughly explored by FTIR, fluorescence, TEM, PXRD, NMR, TGA and Mass spectrometry measurements. The availability of –COOH and –NH functional groups on the surface of the nanofibers has been shown to provide favourable chemical environments for the formation of ultra-thin and uniform sized (∼3 nm) gold nanoparticles (AuNPs). AuNPs embedded in CPH gel matrix (AuCPH) were also found useful as nanocatalysts in the reduction of p-nitrophenol by mild reducing agents like NaBH4. This effect was brought to light with an estimation of the rate constant and activation energy of the reduction reaction estimated at 0.223 min−1/58.94 KJ mol−1 in the presence of AuCPH and at 0.014 min−1/595.14 KJ mol−1, in the absence of AuCPH. CPH as well as dried AuCPH were unable to catalyze the same reaction under similar conditions. Conductivity values reaching 5.05 × 10−3 S cm−1 and 4.18 × 10−3 S cm−1 at 295 K have been determined for AuNP-doped and AuNP-free metallohydrogels, respectively.


 Please wait while we load your content...
Please wait while we load your content...