An amine-functionalized mesoporous silica-supported PdIr catalyst: boosting room-temperature hydrogen generation from formic acid†
Abstract
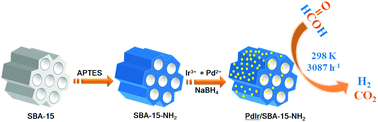
Selective dehydrogenation of formic acid (FA, HCOOH) is a promising alternative to fossil fuels, to provide a clean energy carrier for the future energy economy. The preparation of highly efficient catalysts for hydrogen release from FA at room temperature has attracted much attention but still remains a great challenge. Herein, ultrafine bimetallic PdIr nanoparticles (NPs) immobilized by amine-functionalized SBA-15 (PdIr/SBA-15-NH2) have been successfully synthesized via a facile surface functionalization and co-reduction method. The characterized results showed that ultrafine bimetallic PdIr NPs with a small size of around 1.1 nm were highly dispersed on SBA-15-NH2. Among all the as-synthesized catalysts, the optimized Pd0.85Ir0.15/SBA-15-NH2 nanocomposites (NCs) exhibited the highest catalytic activity and 100% H2 selectivity toward the selective dehydrogenation of FA, giving an initial turnover frequency (TOF) value as high as 3087 h−1 at room temperature. To the best of our knowledge, this is the first report showing that the Ir-containing heterogeneous catalyst can achieve a complete dehydrogenation of FA to H2. The excellent catalytic performance of this catalyst might be attributed to the modified electronic effects of Pd with Ir, the ultrafine size and high dispersion of PdIr NPs, and the synergetic interaction between the PdIr NPs and SBA-15-NH2. The amine functional groups of SBA-15-NH2 not only can lead to ultrafine and well-dispersed PdIr NPs and stabilize the PdIr NPs, but also can serve as Brønsted basic sites, which facilitates the O–H bond dissociation of FA and forms a formate intermediate. The present PdIr/SBA-15-NH2 NCs with efficient catalytic effects on the dehydrogenation of FA may greatly promote the practical application of the FA system on fuel cells.


 Please wait while we load your content...
Please wait while we load your content...