Hydrothermally synthesized chalcopyrite platelets as an electrode material for symmetric supercapacitors†
Abstract
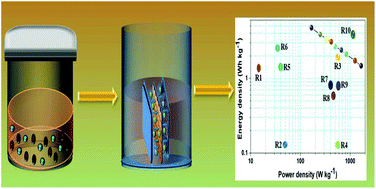
In this work, a novel chalcopyrite (CuFeS2) platelet like open-pored micro-flower structured electrode material was synthesized via a one-step hydrothermal method and its electrochemical performance as an electrode material for supercapacitors were investigated. First and foremost, the structural, morphological, vibrational, and chemical compositional characteristics of the as prepared CuFeS2 were investigated by X-ray diffraction (XRD), field emission scanning electron microscopy (FE-SEM) with elemental mapping, laser Raman spectroscopy, and X-ray photoelectron spectroscopy (XPS), respectively. Subsequently, the electrochemical properties of the CuFeS2 electrode were explored using cyclic voltammetry (CV), galvanostatic charge–discharge (CD), and electrochemical impedance spectroscopy (EIS) studies in 1 M LiOH electrolyte. Cyclic voltammetry and charge–discharge analysis reveal the pseudocapacitive nature of the CuFeS2 electrode by obtaining a maximum specific capacity of about 26.46 mA h g−1 (specific capacitance of about ∼95.28 F g−1) at a scan rate of 5 mV s−1 with a cycling stability retention of 94.38% even after 2000 cycles at a discharge current rate of 5 mA. Furthermore, in view of practical application a symmetric supercapacitor device was fabricated using the CuFeS2 electrode which delivered a maximum specific capacitance of about 34.18 F g−1 at a current rate of 1 mA and a maximum energy density of about 4.74 W h kg−1 with excellent cycling stability. The acquired results confirmed that the CuFeS2 electrode could be a prospective and electrochemically active candidate for next generation supercapacitors.


 Please wait while we load your content...
Please wait while we load your content...