Novel cage-like MOF for gas separation, CO2 conversion and selective adsorption of an organic dye†
Abstract
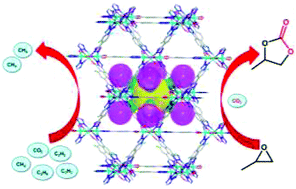
By incorporating the F substituent in tetrazolate–carboxylate ligands and combining the dipyridyl co-linkers, novel Zn-MOF, [(CH3)2NH2][Zn1.5(μ3-O)0.5(F-tzba)1.25(bpy)0.25(μ2-F)0.5]·2DMF·2H2O (1) (F-H2tzba = 2-F-4-(1H-tetrazol-5-yl) benzoic acid, bpy = 4,4′-bipyridine), was constructed. 1 was based on an unprecedented 12-connected hexanuclear cluster and formed an fcu topological framework, which contained intriguing 3D polar pores formed by intersected octahedral and tetrahedral cages with the modification using F. The framework not only showed high adsorption capacity for CO2, C2H2, and C2H6 but also exhibited excellent selective capture for CO2, C2H2, and C2H6 over CH4 and C2H2 over CO2 at 298 K. The framework as a Lewis acid heterogeneous catalyst also displayed highly efficient catalytic activity in the cycloaddition reaction of CO2 with different epoxides to produce cyclic carbonate chemicals. Grand canonical Monte Carlo simulations were studied to identify the different CO2, C2H2 and C2H6 binding sites as well as the catalytic interactions between the framework and the propylene oxide substrate. Simultaneously, the MOF could selectively adsorb the methylene blue (MB) dye from other organic dyes.


 Please wait while we load your content...
Please wait while we load your content...