Understanding the roles of amorphous domains and oxygen-containing groups of nitrogen-doped carbon in oxygen reduction catalysis: toward superior activity†
Abstract
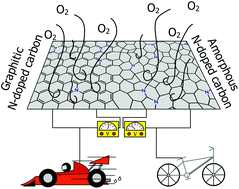
Nitrogen-doped carbons are promising candidates for replacing platinum catalysts in fuel cell electrodes. They typically contain graphitic and amorphous domains, both of which contribute to the oxygen reduction reaction (ORR) activity. Here, we aim at revealing the catalytic functions of each domain as well of the surface functional groups, and ultimately at maximizing the catalytic performance of the materials in the ORR. We develop a sequential oxidative-pyrolytic treatment to remove the amorphous domains and to alter the surface functionalities. The effectiveness of this approach is evidenced by various techniques. Our electrochemical results show a positive correlation between the ORR activity and the degree of graphitization/oxygen functional group removal. While the oxidation-induced oxygen-containing group impairs the ORR, the amorphous domains seem facilitate the 2-electron transfer ORR, lack electrical conductivity and are rich in oxygen-containing groups. The ORR activity of the optimized sample increases considerably in the alkaline electrolyte, giving a half-wave potential >0.85 V (vs. a reversible hydrogen electrode) that is comparable to that of commercial platinum on carbon.


 Please wait while we load your content...
Please wait while we load your content...