Probing the glycopolymer–ion interaction via specific ion effects†
Abstract
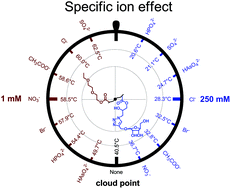
Ions are known to play vital roles in the specific recognition between glycopolymers and lectins; however, it is challenging to characterize such interactions. Herein, specific ion effects are used to probe the interactions between glycopolymers and ions. The effects of different anions and cations on the cloud points of thermoresponsive glycopolymers were investigated. Specific ion effects were found to be pronounced even under low salt concentrations for nonlinear poly(ethylene glycol) (PEG) analogues. The anion's order of the salting-in to salting-out effect for nonlinear PEG analogues was in coincidence with the traditional Hofmeister series. However, due to the presence of high-polarity hydroxyl groups from saccharides, unexpected salting-in effects were observed for the glycopolymers under relatively low concentrations of anions. Both salting-in and salting-out effects could be observed when the glycopolymer was solubilized in aqueous solutions containing low- or high-concentration heavy metal salts. The direct binding of ions to the saccharide units could significantly change the polymers’ hydrophilicity and cloud points, which highlighted the importance of hydroxyl groups in the ionic effects of polymers. The specific ion effect of thermoresponsive glycopolymers could act as a facile tool to study the interactions between saccharides and ions.


 Please wait while we load your content...
Please wait while we load your content...