Impact of amino acids on the aqueous self-assembly of benzenetrispeptides into supramolecular polymer bottlebrushes†
Abstract
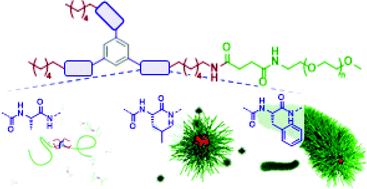
In contrast to covalent polymer brushes, directional supramolecular forces such as hydrogen bonds or π–π-interactions govern the formation of supramolecular polymer bottlebrushes (SPBs) in a self-assembly process starting from single polymer building units. In an attempt to broaden our understanding of these processes and the required supramolecular forces, we here investigated the benzenetrispeptide (BTP) motif to self-assemble polyethylene oxide (PEO) chains into SPBs in water. For this purpose, we synthesized a library of BTP-PEO conjugates, in which we varied the amino acid unit (alanine vs. leucine vs. phenylalanine), the hydrophobic spacer (C6vs. C12), and the steric hindrance imposed by PEO (2 vs. 5 vs. 10 kg mol−1, one vs. three polymer chains). In particular, the type of amino acid was hypothesized to have a crucial influence on the final morphology. A detailed characterization of the respective solutions revealed that for the C6 spacer and one PEO2k chain, only phenylalanine containing core units formed the desirable SPBs, while in the other cases only spherical micelles (leucine) or barely any aggregation (alanine) was observed. In contrast, the more hydrophobic C12 spacer resulted in an exclusive formation of SPBs, whereas the choice of the amino acid moiety had a minor influence on the observed morphology. Increasing the steric hindrance finally resulted in a transition from cylindrical toward spherical micelles. The broad structural variety offered by the choice of amino acids not only allows for an excellent control of the solution morphologies but also the potential integration of functional units to the core.


 Please wait while we load your content...
Please wait while we load your content...